Distilled Water vs Deionized Water
Difference Between Distilled and Deionized Water
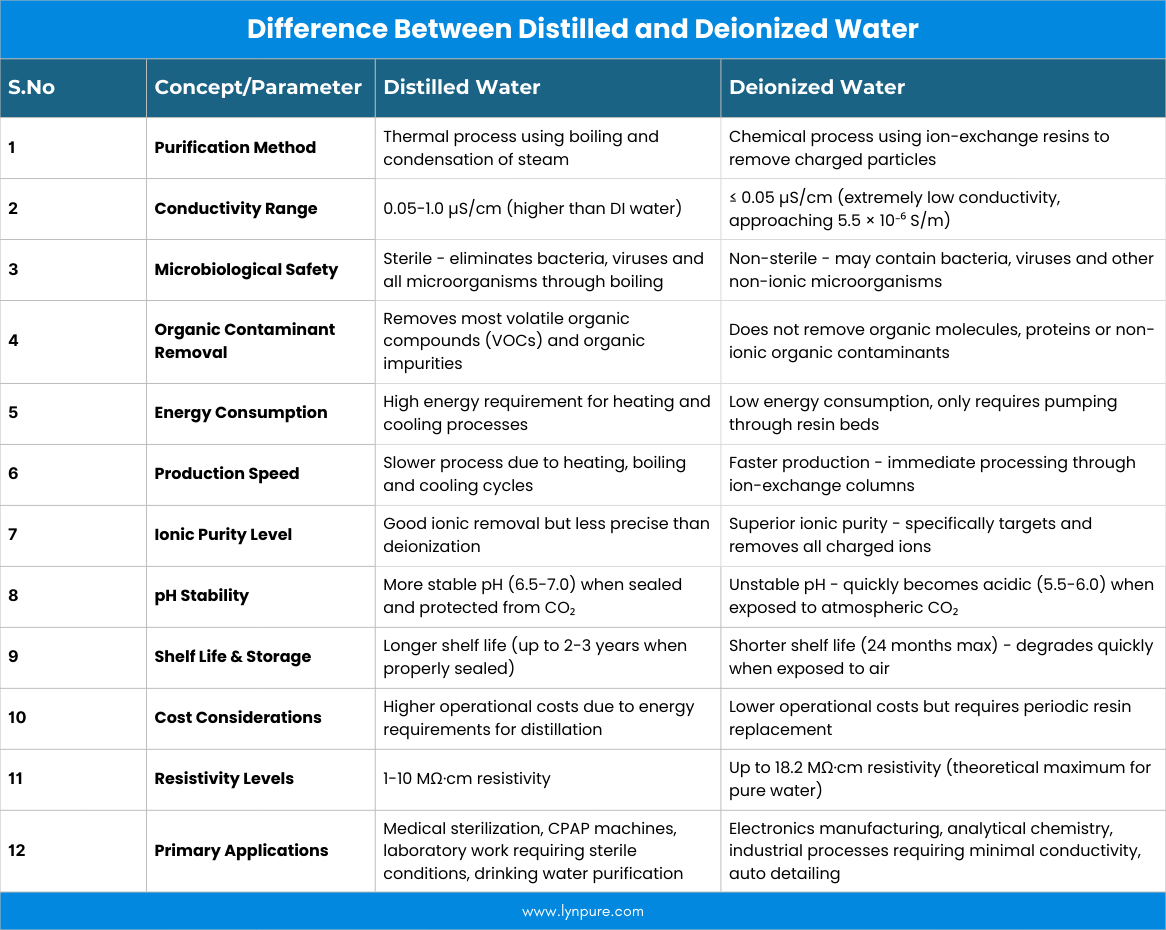
Distilled water is purified by boiling and condensing steam, removing most inorganic salts, minerals and some organic impurities, while deionized (DI) water is purified by passing water through ion-exchange resins that remove all ionic compounds. Distilled water offers higher purity, removing non-ionic substances like bacteria and some volatile organic compounds (VOCs), whereas deionized water excels at eliminating ionic impurities but may retain non-ionic contaminants.
The choice depends on the application: distilled water is safer for drinking and sensitive equipment like CPAP machines, while DI water is preferred for high-tech applications and industrial processes where ionic conductivity must be minimized.
Best For: Laboratories, electronics manufacturing, pharmaceuticals and industrial processes where the absence of ions is critical.
Understanding Water Purification Methods
When comparing different water purification technologies, it's essential to understand that normal water contains various dissolved minerals organic compounds and microorganisms. Each purification method targets specific contaminants, making some more suitable for particular applications than others.
Distilled Water Process:
Water is heated to create steam, which then cools and condenses back into purified water, leaving behind non-volatile impurities. This process removes,
- Minerals
- Salts
- heavy metals
- most organic compounds
- microorganisms.
It's best for drinking water (though not recommended due to lack of minerals), CPAP machines, humidifiers, car batteries and other household appliances where mineral buildup is a concern.
Deionized (DI) Water Process:
Water passes through special resins that attract and remove charged ions, replacing them with hydrogen and hydroxyl ions. This removes ionic impurities like salts and minerals but does not remove non-ionic contaminants like bacteria, viruses and organic molecules. It's best for laboratories, electronics manufacturing, pharmaceuticals and industrial processes where the absence of ions is critical.
How Is Distilled Water Made: The Complete Process
The distillation process involves several key stages that ensure maximum purity. First, normal water is heated in a boiling chamber until it reaches its boiling point, converting liquid water into steam. This steam rises and travels through a cooling system where it condenses back into liquid form, now as condensed water free from most impurities. During this transformation, heavy metals, dissolved minerals, bacteria and most organic compounds remain in the original boiling chamber, unable to vaporize at water's boiling point. The resulting distilled water typically has a purity level of 99.9%, making it significantly cleaner than filtered water or RO water systems.
Deionization Process:
Advanced Ion Exchange Technology
The deionization process relies on sophisticated ion exchange resins that selectively remove charged particles from water. These specialized resins contain either positively charged sites (cation exchange resins) or negatively charged sites (anion exchange resins) that attract and bind opposite-charged ions from the water.
What is ion exchange exactly?
It's a chemical process where unwanted ions in the water are replaced with more desirable ones. Cation resins replace positive ions like sodium, calcium and magnesium with hydrogen ions, while anion resins replace negative ions like chloride and sulfate with hydroxyl ions. When hydrogen and hydroxyl ions combine, they form pure water molecules.
Boiling Water vs Distilled Water: Key Differences
Many people wonder about the difference between simply boiling water and creating distilled water. While both processes involve heating water, they serve different purposes and achieve different results. Boiling water primarily kills bacteria, viruses and other microorganisms, making it safer to drink. However, boiled water still contains all the original dissolved minerals, salts and other impurities. The boiling process doesn't remove these substances; it only eliminates biological contaminants.
In contrast, distilled water goes beyond simple boiling. The distillation process not only kills microorganisms but also physically separates water from dissolved impurities through evaporation and condensation. This makes distilled water significantly purer than merely boiled water.
Comparing Water Types: Which Is Best for Your Needs?
Drinking Water Applications
For drinking water purposes, both distilled water and deionized water have limitations. While these purified waters are safe to consume, they lack essential minerals that contribute to taste and health benefits. Many people prefer filtered water or RO water for drinking, as these methods remove harmful contaminants while retaining beneficial minerals. Salt water, obviously, requires extensive purification before consumption. Both distillation and reverse osmosis can effectively desalinate salt water, though distillation is more energy-intensive for large-scale operations.
Industrial and Laboratory Uses
Deionized water excels in applications requiring minimal electrical conductivity, such as:
- Electronics manufacturing and cleaning
- Laboratory experiments and analysis
- Pharmaceutical production
- Steam boilers and cooling systems
Distilled water proves superior when complete purity is needed, including:
- Medical device sterilization
- Battery maintenance and manufacturing
- Research requiring contaminant-free water
- Precision cleaning applications
The Science Behind Water Purity Levels
Understanding purity measurements helps determine which water type suits your needs. Distilled water typically achieves total dissolved solids (TDS) levels below 10 parts per million (ppm), while deionized water can reach even lower ionic content but may contain higher levels of organic compounds. Normal water from municipal sources usually contains 100-300 ppm TDS, filtered water ranges from 50-150 ppm depending on the filtration method and RO water typically achieves 5-25 ppm TDS levels.
The Role of RO Water in Advanced Water Purification Systems
While distilled water and deionized water represent distinct purification methods, they often work in conjunction with reverse osmosis (RO) water systems to achieve superior results. It's important to understand that neither distilled nor deionized water is considered RO water, as each employs different purification mechanisms. However, RO water purification serves as an excellent pre-treatment step for both distillation and deionization processes.
Understanding RO Water Purification
RO water purification uses a semi-permeable membrane to remove contaminants by applying pressure to force water molecules through microscopic pores while blocking larger particles, dissolved salts, bacteria and organic compounds. This water purification method typically removes 95-99% of total dissolved solids, producing high-quality water that serves as an ideal starting point for further purification.
Combining RO with Distillation and Deionization
Many industrial and laboratory facilities use RO water as feedwater for distillation and deionization systems. Starting with RO-purified water offers several advantages:
For Distillation Systems:
- Reduces energy consumption by eliminating most contaminants before the heating process
- Minimizes scale buildup in distillation equipment
- Extends equipment lifespan and reduces maintenance requirements
- Improves final product quality and consistency
For Deionization Systems:
- Significantly extends ion exchange resin life
- Reduces operational costs through less frequent resin replacement
- Prevents organic fouling of ion exchange resins
- Ensures more consistent deionized water quality
Multi-Stage Water Purification Approaches
Progressive water purification systems often combine multiple technologies in sequence. A typical high-purity water system might include:
- Pre-filtration to remove sediments and chlorine
- RO water purification to eliminate most dissolved contaminants
- Deionization for final ionic impurity removal
- UV sterilization for microbiological control
This multi-stage approach produces ultra-pure water exceeding the quality achievable by any single purification method. Such systems are common in pharmaceutical manufacturing, semiconductor production and critical research applications where water purity directly impacts product quality and experimental results.
When RO Water Alone Is Sufficient
For many applications, RO water provides adequate purity without requiring additional distillation or deionization. These include:
- General laboratory use where moderate purity suffices
- Food and beverage production
- Aquaculture and hydroponic systems
- General industrial processes with less stringent purity requirements
Understanding when RO water meets your needs versus when you require the enhanced purity of distilled water or deionized water helps optimize both costs and performance in your specific application.
Cost and Practical Considerations
Both distilled water and deionized water production require specialized equipment and ongoing operational costs. Distillation demands significant energy for heating and cooling, making it more expensive for large-volume applications. The deionization process requires periodic resin replacement but uses less energy during operation. For home use, purchasing distilled water is often more economical than investing in distillation equipment. However, for businesses requiring large quantities of purified water, installing appropriate systems may prove more cost-effective long-term.
Environmental Impact and Sustainability
When choosing between water purification methods, consider environmental factors. Distillation requires substantial energy input, contributing to higher carbon footprints. Deionization generates less heat waste but produces spent resins that require proper disposal. RO water systems and advanced filtered water technologies often provide good compromises between purity levels and environmental impact, making them popular choices for both residential and commercial applications.
Maintenance and Storage Requirements
Both distilled water and deionized water require careful handling to maintain their purity. These purified waters can quickly absorb contaminants from the air, containers and handling equipment. Proper storage in clean, sealed containers prevents recontamination. Regular maintenance of production equipment ensures consistent water quality. Distillation systems need periodic cleaning to remove mineral deposits, while deionization systems require resin replacement based on usage levels and water quality.
Making the Right Choice for Your Application
The decision between distilled water and deionized water ultimately depends on your specific requirements. Consider these factors:
- Purity needs: Choose distilled water for maximum overall purity, deionized water for ionic purity.
- Budget constraints: Evaluate both initial equipment costs and ongoing operational expenses.
- Volume requirements: Large-scale operations may favor one method over another.
- Application sensitivity: Some processes specifically require ion-free water, while others need complete contaminant removal.
Understanding these differences ensures you select the most appropriate water purification method for your needs, whether for industrial processes, laboratory work or specialized household applications.